Online Health Economic Models with PharmEconix©
To provide a strong infrastructure for good modeling practice considering:
1. Clinical realism: A model should reflect the state of evidence, the current understanding of the disease, and be accepted by clinical experts.
2. Quantifying decision uncertainty: A model should be capable of quantifying decision uncertainty and informing prioritization of future research.
3. Transparency and reproducibility: Resources should exist so that a model can be completely understood, reproduced, and pressure tested.
4. Reusability and adaptability: It should be possible to easily update a model to reflect new clinical evidence or adapt it for a new market, indication, or intervention.
Reference: https://doi.org/10.1016/j.jval.2019.01.003
To support our clients for a more efficient introduction of new products to their target groups such as policymakers, clinicians, and patient organizations.
To provide a quick updating process for the economic model when a new set of input data are available.
To enhance model adaptation for multiple countries all in one platform.
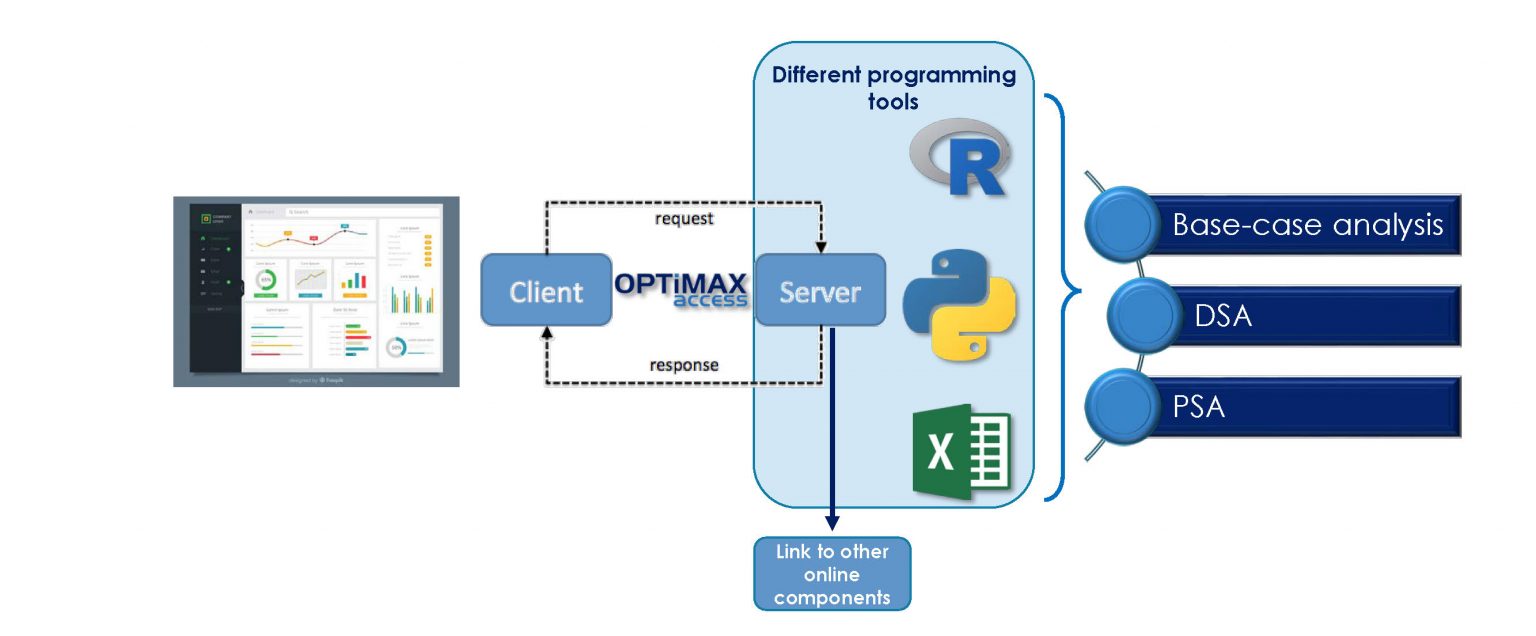
Architecture
The system consists of two main parts: server-side and client-side. Model engines are developed with the support of R, Python, or Excel on the server-side.
Depending on the values users submit to the system, the server-side runs the analysis and generates the results. The client-side provides a graphical user interface (GUI) for users to interact with the platform and make appropriate adjustments to the model settings and input values.
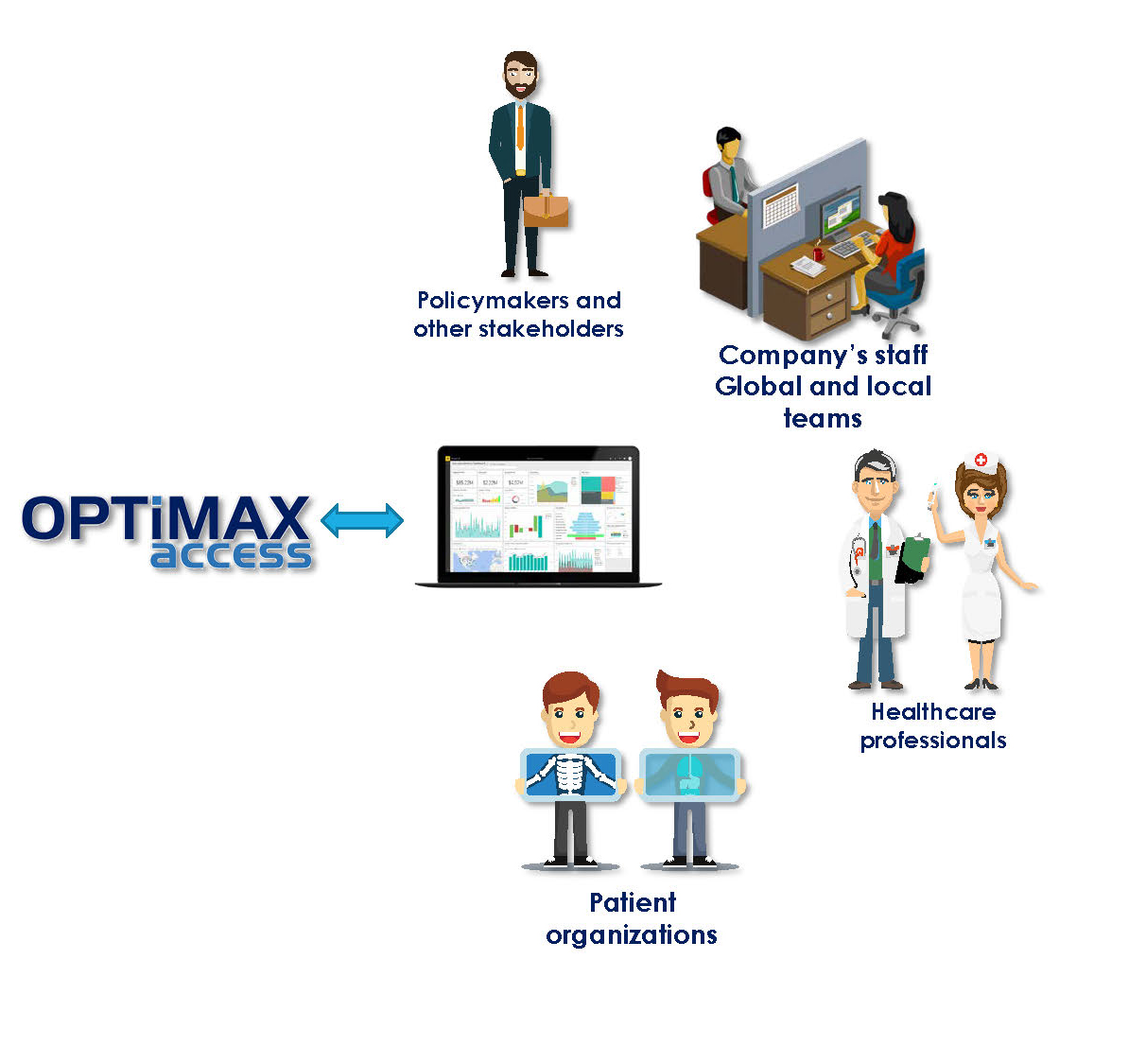
The system has a model administrator to oversee its core-level features. It also supports several user types who can work with the model on the online platform. The main supported user types are local and global team members of the product manufacturers, local policymakers, medical providers, and patient organizations. The model admin can define the level of access given to each user to ensure system security.
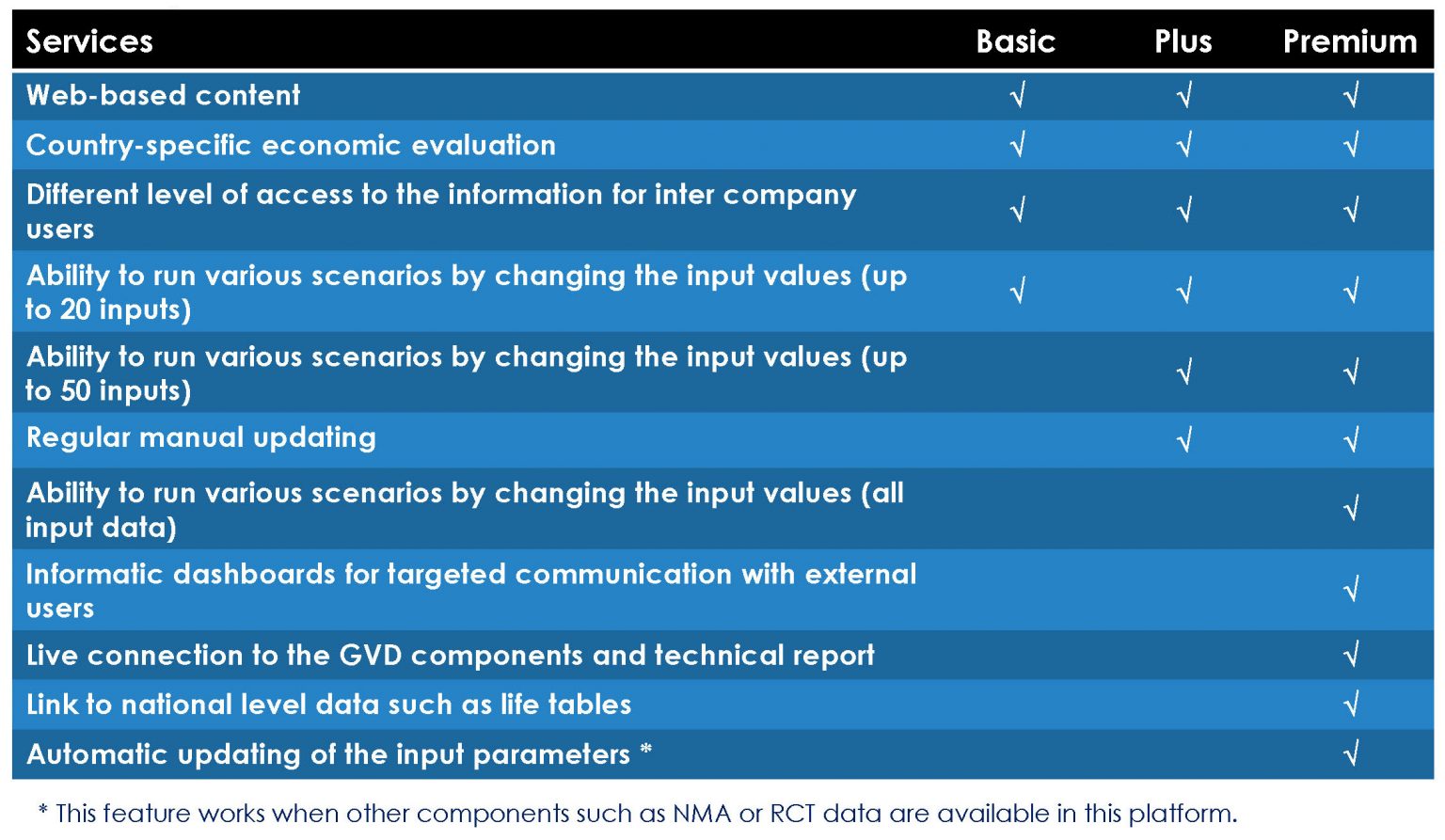
- Different Packages for the Online Model
OUR SERVICES
Have Questions?
Please complete the form below to contact us for more information